Raoult's law
The Raoult's law relates PA in the gas and XA in the liquid.
YAP=XAP*A
When the composition of liquid XA=1
Where:
YA=gas composition
P=system pressure
XA=liquid composition
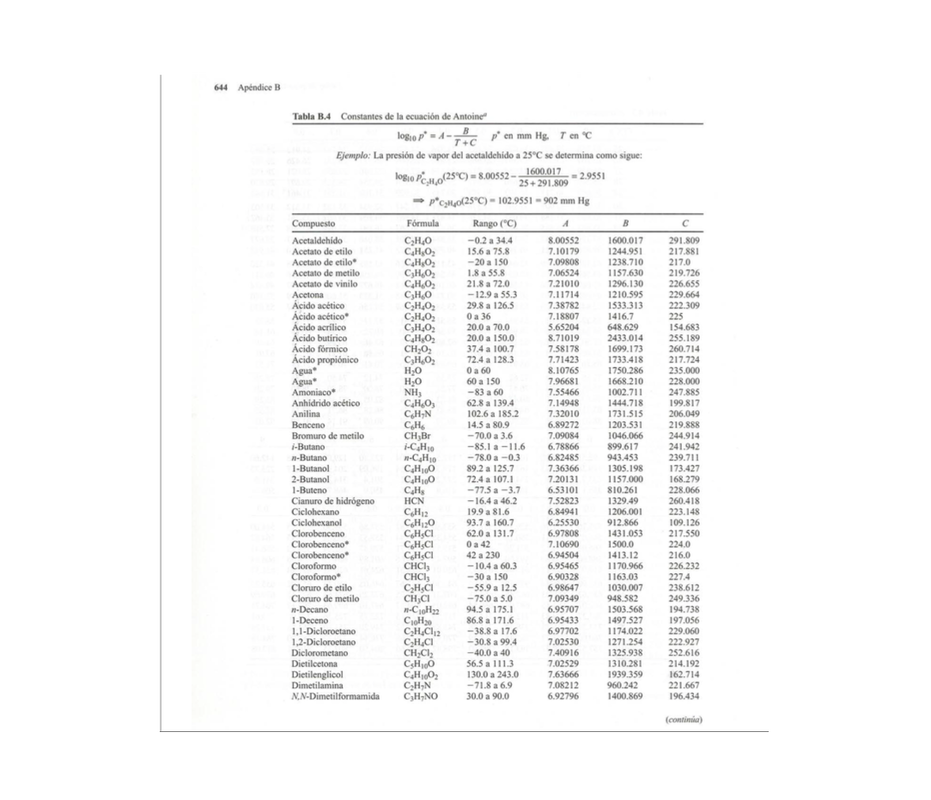
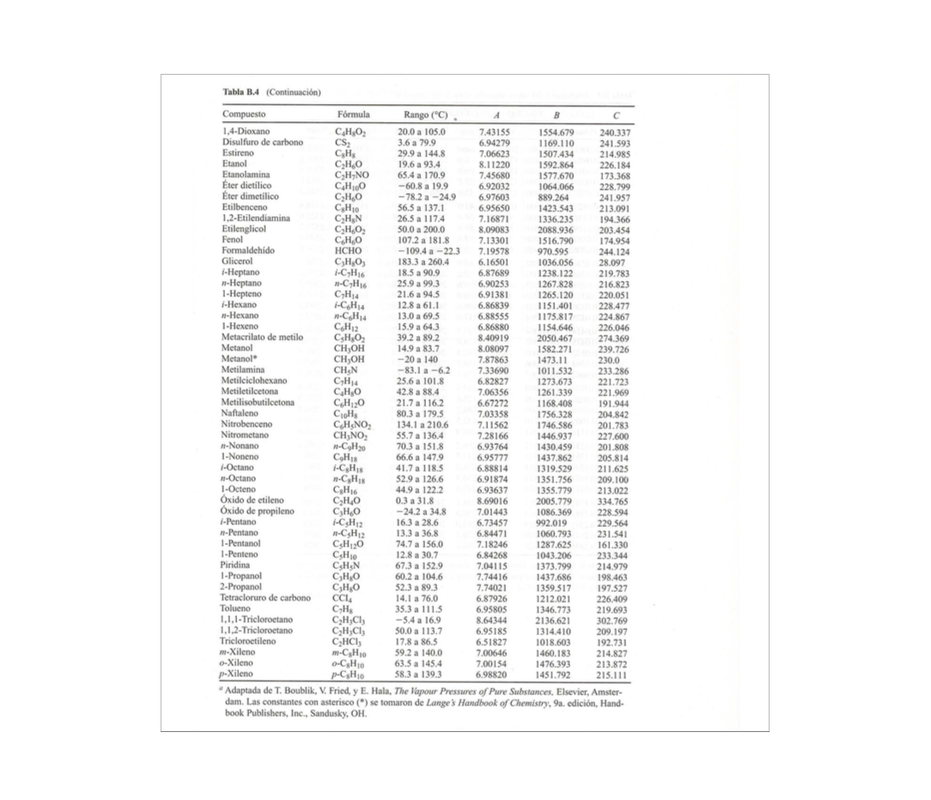
PA*=Antoine Law Antoine Law P*=10^(A+(B/C+T)
A, B and C are experimental constants that you can find in the following tables.
Aplication.
RAOULT'S LAW-BASED METHOD FOR DETERMINATION OF COAL TAR AVERAGE MOLECULAR WEIGHT
A Raoult's law-based method for determining the number average molecular weight of coal tars is presented. The method requires data from two-phase coal tar/water equilibrium experiments, which readily are performed in environmental laboratories. An advantage of this method for environmental samples is that it is not impacted by the small amount of inert debris often present in coal tar samples obtained from contaminated sites. Results are presented for 10 coal tars from nine former manufactured gas plants located in the eastern United States. Vapor pressure osmometry (VPO) analysis provided similar average molecular weights to those determined with the Raoult's law-based method, except for one highly viscous coal tar sample. Use of the VPO-based average molecular weight for this coal tar resulted in underprediction of the coal tar constituents' aqueous concentrations. Additionally, one other coal tar was not completely soluble in solvents used for VPO analysis. The results indicate that the Raoult's law-based method is able to provide an average molecular weight that is consistent with the intended application of the data (e.g., modeling the dissolution of coal tar constituents into surrounding waters), and this method can be applied to coal tars that may be incompatible with other commonly used methods for determining average molecular weight, such as vapor pressure osmometry.
RAOULT'S LAW-BASED METHOD FOR DETERMINATION OF COAL TAR AVERAGE MOLECULAR WEIGHT
A Raoult's law-based method for determining the number average molecular weight of coal tars is presented. The method requires data from two-phase coal tar/water equilibrium experiments, which readily are performed in environmental laboratories. An advantage of this method for environmental samples is that it is not impacted by the small amount of inert debris often present in coal tar samples obtained from contaminated sites. Results are presented for 10 coal tars from nine former manufactured gas plants located in the eastern United States. Vapor pressure osmometry (VPO) analysis provided similar average molecular weights to those determined with the Raoult's law-based method, except for one highly viscous coal tar sample. Use of the VPO-based average molecular weight for this coal tar resulted in underprediction of the coal tar constituents' aqueous concentrations. Additionally, one other coal tar was not completely soluble in solvents used for VPO analysis. The results indicate that the Raoult's law-based method is able to provide an average molecular weight that is consistent with the intended application of the data (e.g., modeling the dissolution of coal tar constituents into surrounding waters), and this method can be applied to coal tars that may be incompatible with other commonly used methods for determining average molecular weight, such as vapor pressure osmometry.
References.
- LearnCemE (2012). Raoult's Law Explanation. Retrieved from: http://youtu.be/Adr9_2LnQdw
- Brown, D. G., Gupta, L., Moo-Young, H., & Coleman, A. J. (2005). RAOULT'S LAW-BASED METHOD FOR DETERMINATION OF COAL TAR AVERAGE MOLECULAR WEIGHT. Environmental Toxicology and Chemistry, 24(8), 1886-92. Retrieved from http://0-search.proquest.com.millenium.itesm.mx/docview/210342551?accountid=11643
- LearnCemE (2012). Raoult's Law Explanation. Retrieved from: http://youtu.be/Adr9_2LnQdw
- Brown, D. G., Gupta, L., Moo-Young, H., & Coleman, A. J. (2005). RAOULT'S LAW-BASED METHOD FOR DETERMINATION OF COAL TAR AVERAGE MOLECULAR WEIGHT. Environmental Toxicology and Chemistry, 24(8), 1886-92. Retrieved from http://0-search.proquest.com.millenium.itesm.mx/docview/210342551?accountid=11643