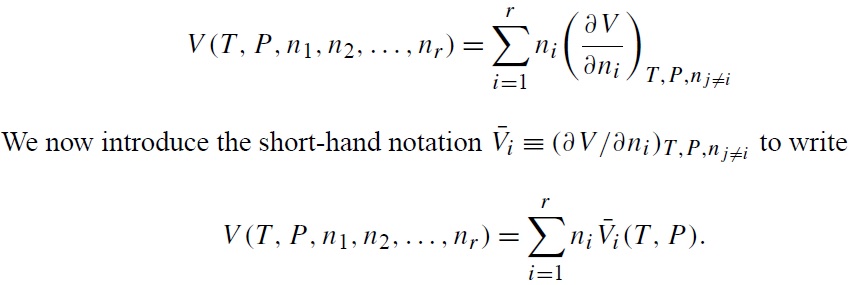Partial molar properties
Remembering the phase rule of a pure substance
F=C-P+2 C=1 P=1
F=2
We need to specify 2 intensive variables to describe a single phase system.
Extensive properties: Specify total number of moles
K =Extensive thermodynamic property of entire mixture (V, H, U, S, G)
k=Intensive property: K/#moles (v, h, u, s, g)
K can be written as the sum of partial derivates of each independent variable:
dK=(∂K/∂T)P,ni dT+ (∂K/∂P)T,ni
dP +∑(∂K/∂ni)P,T,nj=1 dni
¯Ki define any partial molar property
¯Ki= (∂K/∂ni)P,T,nj=1 dni
If the composition is kept constant, the partial molar propertie are constant.
K=∑¯Ki*ni+C C=0 as ∑ni=0
So
V=∑niVi H=∑niHi
Example: Partial Molar Volumes
The volume of a system depends on its chemical composition in a mixture for wich the mole numbers of the constituent components are specified. Consider as an example an aqueous solution containing sulfuric acid and sodium chloride at constant temperatura and pressure. If we double the mole numbers of each component, we doublé the volume of the system. Thus, more generally, at fixed T and P the volume of a system should be a homogeneous funtion of the mole numbers ni of all species in the system, independent of the state of aggregation of each constituent. Then, consider the next equation.
The volume of a system depends on its chemical composition in a mixture for wich the mole numbers of the constituent components are specified. Consider as an example an aqueous solution containing sulfuric acid and sodium chloride at constant temperatura and pressure. If we double the mole numbers of each component, we doublé the volume of the system. Thus, more generally, at fixed T and P the volume of a system should be a homogeneous funtion of the mole numbers ni of all species in the system, independent of the state of aggregation of each constituent. Then, consider the next equation.
Considerations of the Partial Molar Properties.
-Species become part of a mixture, it losses its identity.
-Each species contribute to the property of the mixture.
-Total solution property depend on amount of each species.
-Species become part of a mixture, it losses its identity.
-Each species contribute to the property of the mixture.
-Total solution property depend on amount of each species.
References.
Honig, J. M. (2007). Thermodynamics : Principles Characterizing Physical and Chemical Processes. Amsterdam: Academic Press.
Honig, J. M. (2007). Thermodynamics : Principles Characterizing Physical and Chemical Processes. Amsterdam: Academic Press.