Equilibrium in multicomponent systems
For understand better the equilibrium in multicomponent systems we have to know the following:
-Chemistry: mixtures that react
-General concepts of mixtures
-First step: mixtures that do not react, mainly binary (XA+XB=1)
-Mixtures: more complex than pure substances in terms of intermolecular reactions
-Chemistry: mixtures that react
-General concepts of mixtures
-First step: mixtures that do not react, mainly binary (XA+XB=1)
-Mixtures: more complex than pure substances in terms of intermolecular reactions
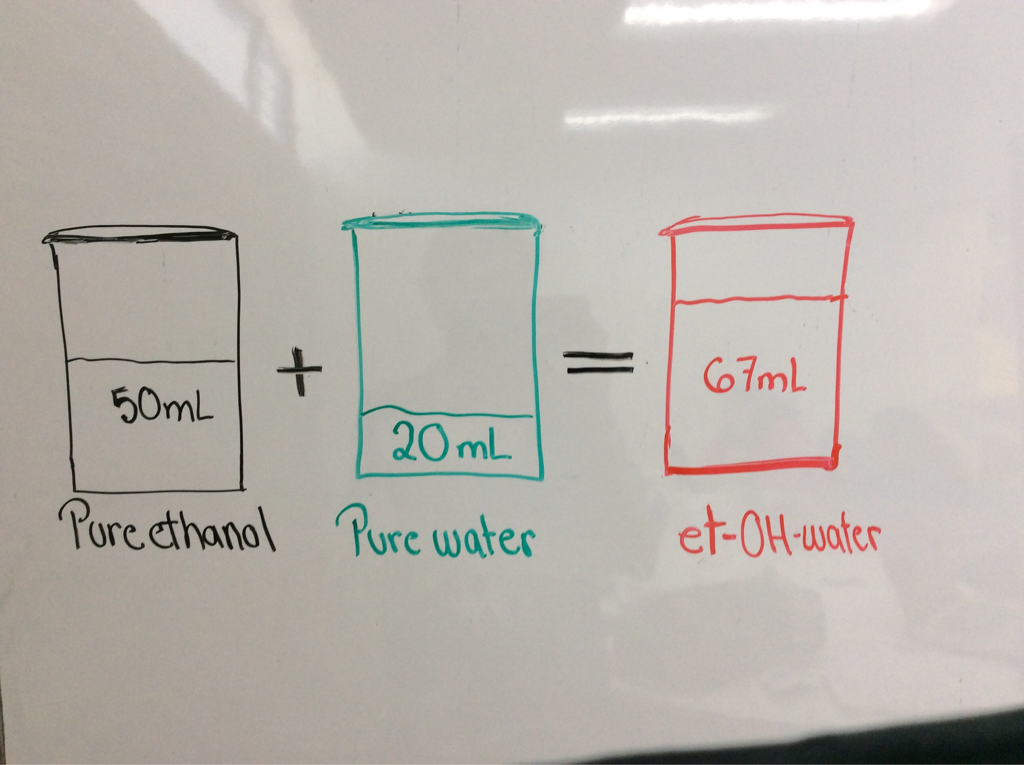
In a container there are 50 mL of pure ethanol, and in another one there are 20 mL of pure water. When they are mixed, and create a solution of 67 mL, so. What happened with the 3mL?
This happend because of the interactions of the molecules like hydrogen bonds, the volume change but the mass is preseved.
First we have to know some things.
-Species become part of a mixture and loose their identities.
-Each species contribute to the properties of mixtures.
-The total solution properties depend on the amount of each species.
In the last mixture, the ethanol molecules are much smaller than water's. The smaller molecules tend to cram between the bigger ones, and it generates a smaller volume than what is expected.
This happend because of the interactions of the molecules like hydrogen bonds, the volume change but the mass is preseved.
First we have to know some things.
-Species become part of a mixture and loose their identities.
-Each species contribute to the properties of mixtures.
-The total solution properties depend on the amount of each species.
In the last mixture, the ethanol molecules are much smaller than water's. The smaller molecules tend to cram between the bigger ones, and it generates a smaller volume than what is expected.