Entropy.
One of the ideas involved in the concept of entropy is that nature tends from order to disorder in isolated systems.
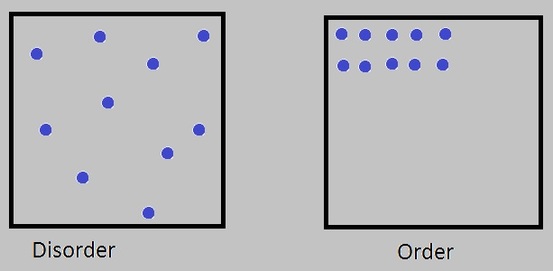
For example, in these two systems, cointaining gas molecules, the most probable order is disorder!
For an isolated system, entropy always increases or remains the same, so if you compare states of different entropy, the one with the greater entropy will be later in time.
Sometimes entropy is hard to understand in terms of order and disorder in chemestry, so is necesary to observe, based in the diagrams shown above, the next information.
At ordinary temperatures, the internal energy of a gas would give the molecules high velocities, and it is evident that this orderly arrangement would be very rare because there are only a few ways to do it. If it occurred, it would be for a brief instant and then the molecules would move to some other configuration. The first diagram depicts a more random or disordered configuration, but the key point is that there is a vast number of ways that such configurations could be achieved. So multiplicity is the key concept - molecular ensembles will spontaneously tend to evolve from configurations of lower multiplicity to configurations of greater multiplicity.
Entropy is a crucial microscopic concept for describing the thermodynamics of systems of molecules. The idea of disorder is more an introductory visualization.
Sometimes entropy is hard to understand in terms of order and disorder in chemestry, so is necesary to observe, based in the diagrams shown above, the next information.
At ordinary temperatures, the internal energy of a gas would give the molecules high velocities, and it is evident that this orderly arrangement would be very rare because there are only a few ways to do it. If it occurred, it would be for a brief instant and then the molecules would move to some other configuration. The first diagram depicts a more random or disordered configuration, but the key point is that there is a vast number of ways that such configurations could be achieved. So multiplicity is the key concept - molecular ensembles will spontaneously tend to evolve from configurations of lower multiplicity to configurations of greater multiplicity.
Entropy is a crucial microscopic concept for describing the thermodynamics of systems of molecules. The idea of disorder is more an introductory visualization.
Entropy and Second Law of Thermodynamics.
In the first law of thermodynamics considers a conservation of mass and energy, but also considers a time/symetric process, something that does not occur in real life. So The Second law expresses which changes are going to occur, what motivates the change, and the direction in wich some event is going.
|
The Second Law of Thermodynamics states that the state of entropy of the entire universe, as a closed isolated system, will always increase over time. The second law also states that the changes in the entropy in the universe can never be negative.
|
For a better unerstandig of the topic, you can see this video.
|
Units
s=KJ/[Kg][K]. SI Unit System.
s=Btu/[lbm][R]. US Unit System.
s=KJ/[Kg][K]. SI Unit System.
s=Btu/[lbm][R]. US Unit System.
References.
CrashCourse (2013). Entropy: Embrace the chaos! Crash Course Chemestry #20. Retrieved from: https://www.youtube.com/watch?v=ZsY4WcQOrfk
Georgia State University (n. d.). Entropy as Time's Arrow. Retrieved from: http://hyperphysics.phy-astr.gsu.edu/hbase/therm/entrop.html
UCDAVID. (n. d.). 2nd Law of Thermodynamics. In Chemwiki. Retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Thermodynamics/Laws_of_Thermodynamics/Second_Law_of_Thermodynamics
CrashCourse (2013). Entropy: Embrace the chaos! Crash Course Chemestry #20. Retrieved from: https://www.youtube.com/watch?v=ZsY4WcQOrfk
Georgia State University (n. d.). Entropy as Time's Arrow. Retrieved from: http://hyperphysics.phy-astr.gsu.edu/hbase/therm/entrop.html
UCDAVID. (n. d.). 2nd Law of Thermodynamics. In Chemwiki. Retrieved from: http://chemwiki.ucdavis.edu/Physical_Chemistry/Thermodynamics/Laws_of_Thermodynamics/Second_Law_of_Thermodynamics