Gibbs free energy
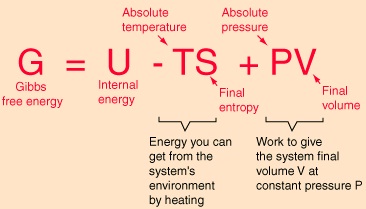
The Gibbs Free Energy is defined by:
The internal energy U might be thought of as the energy required to create a
system in the absence of changes in temperature or volume. An environment at constant temperature T will contribute an amount TS to the
system, reducing the overall investment necessary for creating the system. This
net energy contribution for a system created in environment temperature T from a
negligible initial volume is the Gibbs free energy.
The change in Gibbs free energy, ΔG, in a reaction is a very useful parameter. It can be thought of as the maximum amount of work obtainable from a reaction.
The change in Gibbs free energy, ΔG, in a reaction is a very useful parameter. It can be thought of as the maximum amount of work obtainable from a reaction.
Gibbs Gree Energy and Chemical Reactions.
The change in Gibbs free energy associated with a chemical reaction is a useful indicator of whether the reaction will proceed spontaneously. Since the change in free energy is equal to the maximum useful work which can be accomplished by the reaction. ΔG=Wmax.
then a negative ΔG associated with a reaction indicates that it can happen spontaneously. This is consistent with the usual chemistry convention of treating work done by the system as negative work. Most common reactions can be assessed for spontaneity under standard conditions by looking up the associated thermodynamic quantities for each of the reactants and products.
ΔG=ΔH-TΔS.
The change in Gibbs free energy associated with a chemical reaction is a useful indicator of whether the reaction will proceed spontaneously. Since the change in free energy is equal to the maximum useful work which can be accomplished by the reaction. ΔG=Wmax.
then a negative ΔG associated with a reaction indicates that it can happen spontaneously. This is consistent with the usual chemistry convention of treating work done by the system as negative work. Most common reactions can be assessed for spontaneity under standard conditions by looking up the associated thermodynamic quantities for each of the reactants and products.
ΔG=ΔH-TΔS.
Aplications.
Have you ever wanted to know how much energy do you need to conduct a dissociation? For example this one
H2O2(g) →H2O(g) + ½ O2(g).
Well to answer that question we have the Gibbs Free Energy.
The Gibbs Free Energy is the energy required to do a work and this is its equation:
∆G=∆H-T*∆S
For understand better the Gibbs energy we have an example :
Microbial growth is a biological process that has been previously treated as a chemical reaction operating in accord with the Gibbs free energy equation, developed during the 1870s. However, the full application of the Gibbs equation to microbial growth did not occur until 1997, with the experimental measurement of yeast cell entropy.
Dr. Edwin H. Battley, emeritus of Stony Brook University and recipient of the International Society for Biological Calorimetry's Dubrunfaut Award (1994) and Lavoisier Medal (2010), examined the use of the Gibbs free energy equation to accurately determine the change in energy that accompanies cellular growth.
In many systems, the values for some variables cannot be determined experimentally and so must be calculated from theoretically derived values. The free energy change accompanying cellular growth cannot be directly measured but, if the heat of growth can be measured and the entropy change accompanying growth can be calculated indirectly from heat measurements, the free energy change can be calculated using the Gibbs free energy equation.
Calculate the thermodynamics of microbial growth !
*Using the Gibbs Free Energy you can know if two phases are in equilibrium.
References.
- Hyperphysics.phy (n. d.) Gibbs Free Energy. Retrieved from: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/helmholtz.html#c2
- Bozeman Science (2011). Gibbs Free Energy. Retrieved from: http://youtu.be/DPjMPeU5OeM.
- Bozeman Science (2014). Using Gibbs Free Energy. Retrieved from: http://youtu.be/huKBuShAa1w.
- Hyperphysics.phy (n. d.) Gibbs Free Energy. Retrieved from: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/helmholtz.html#c2
- Bozeman Science (2011). Gibbs Free Energy. Retrieved from: http://youtu.be/DPjMPeU5OeM.
- Bozeman Science (2014). Using Gibbs Free Energy. Retrieved from: http://youtu.be/huKBuShAa1w.