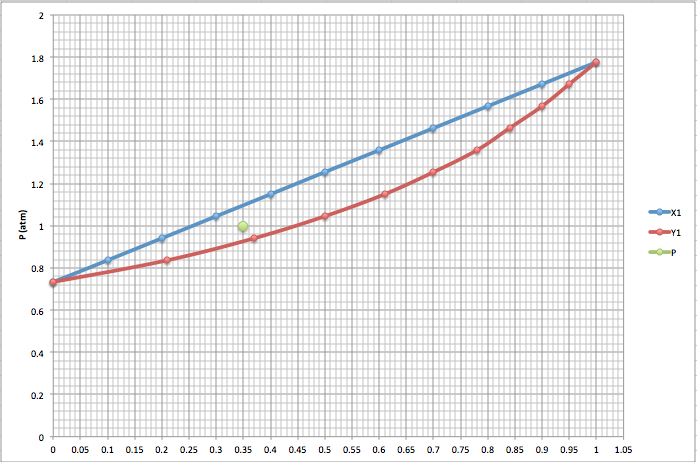
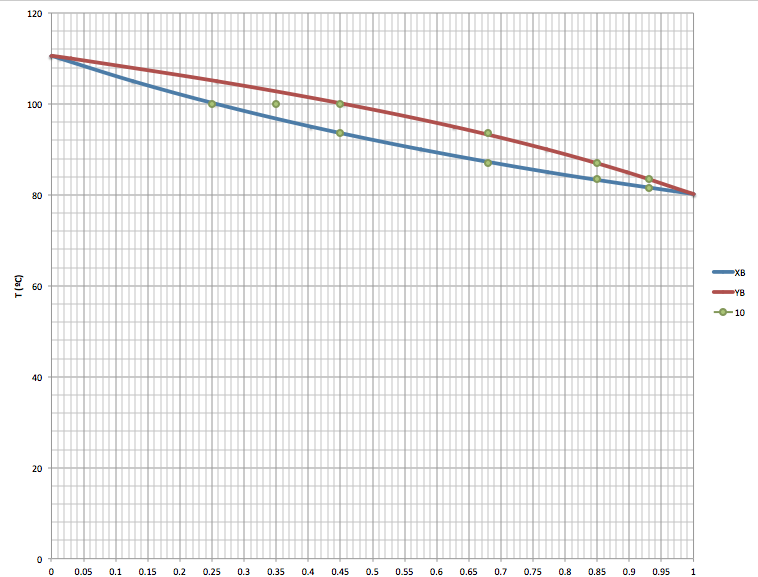
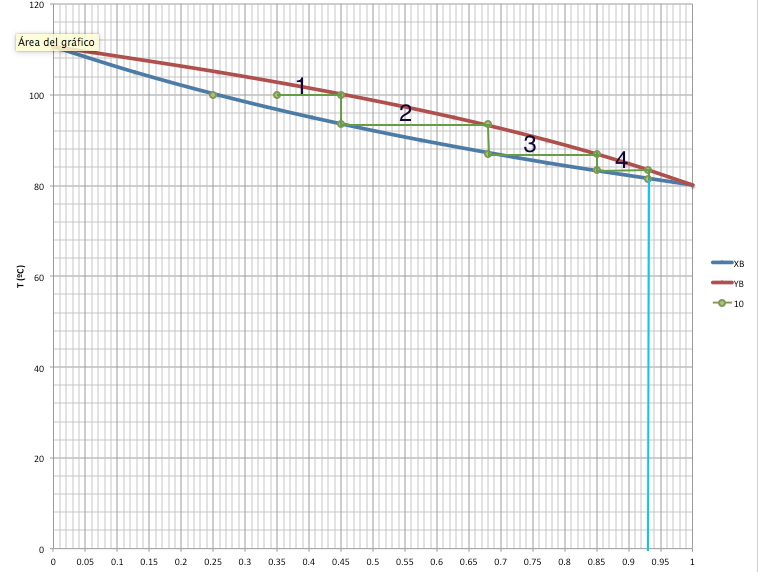
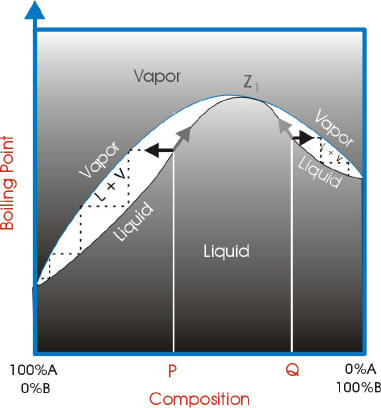
Sometimes we need graphic representations of the binary systems in order to understand better the behaviour of the components. For this situations we can make two types of graphics: P(xy) and T(x,y).
P(x,y): keeping a constant temperature vs concentration of the components (xy).
T(xy): keeping a constant pressure vs concentration of the component (xy)
P(x,y): keeping a constant temperature vs concentration of the components (xy).
T(xy): keeping a constant pressure vs concentration of the component (xy)
This graphics also help us to know the theorical plates that are the number of effective vaporisation and condensation steps that are required to achive a condensate phase of a given composition for a given distillate.
When a mixture of two or more liquids give a vapor that is in equilibrium with its liquid and have the same composition of the liquid, the mixture is called an azeotrope.
Simulations.
|
|
|
References:
- Dupont Durst, H. Gokel, George W.. (2007). Química orgánica experimental. España: Reverté.
- LearnChemE (2015). P-x-y and T-x-y Diagram for VLE (Interactive Simulation). Retrieved from: http://youtu.be/19gr-1ePMAI
- LearnChemE (P-x-y Diagram for VLE (Interactive Simulation). Retrieved from: http://youtu.be/6l3NTuYkeC8
- Dupont Durst, H. Gokel, George W.. (2007). Química orgánica experimental. España: Reverté.
- LearnChemE (2015). P-x-y and T-x-y Diagram for VLE (Interactive Simulation). Retrieved from: http://youtu.be/19gr-1ePMAI
- LearnChemE (P-x-y Diagram for VLE (Interactive Simulation). Retrieved from: http://youtu.be/6l3NTuYkeC8