Phase transition
Definitions
Phase: Uniform form of matter that is homogeneous through at in chemical composition and phisical state.
Phase transition: Spontaneous convertion from one phase to another.
In a closed system
Vapour pressure: pressure of vapour in equilibrium with the condense phase.
L-V boundarie: vapour pressure varies with T
S-V boundarie: variation of sublimation vapour with T
In an open system
Vapour pressure: External P=T (boiling point)
Boiling: free vaporization of liquid
Phase Rule
Number of parameters that can be varied independently while the number of phases in equilibrium is preserved.
F=C-P+2
F= degrees of freedom
C=components
P=phases
For example:
In case of a pure substance:
C=1 P=1
F=1-1+2
F=2
F=C-P+2
F= degrees of freedom
C=components
P=phases
For example:
In case of a pure substance:
C=1 P=1
F=1-1+2
F=2
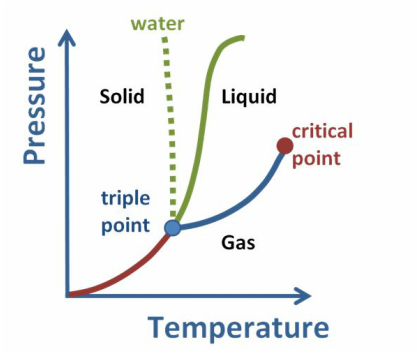
Phase diagram
A phase diagram is map of P and T at which each phase of a substance is more stable.